23262.“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
first law of thermodynamics
second law of thermodynamics
conservation of momentum
conservation of mass
23263.Helium gas goes through a cycle ABCDA (consisting of two isochoric and isobaric lines) as shown in figure. Efficiency of this cycle is nearly
(Assume the gas to be close to ideal gas)
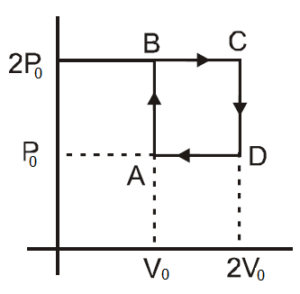
(Assume the gas to be close to ideal gas)

15.4%
9.1%
10.5%
12.5%
23264.A Carnot engine, having an efficiency of η =1/10 as heat engine, is used as a refrigerator. If the work done on the system is 10 J, the amount of energy absorbed from the reservoir at lower temperature is
100 J
99 J
90 J
1 J
23265.The specific heat capacity of a metal at low temperature (T) is given as Cp(kJK–1kg–1) = 32$\left(\dfrac{\text{T}}{400}\right)^3$. A 100 gram vessel of this metal is to be cooled from 20 K to 4 K by a special refrigerator operating at room temperature (27°C). The amount of work required to cool the vessel is
Less than 0.028 kJ
Equal to 0.002 kJ
Greater than 0.148 kJ
Between 0.148 kJ and 0.028 kJ
23266.A Carnot engine working between 300 K and 400 K has 800 J of useful work. The amount of heat energy supplied to the engine from the source is
2400 J
3200 J
1200 J
3600 J
