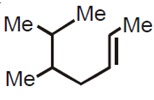
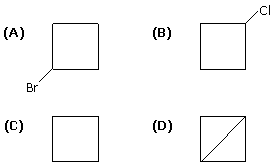
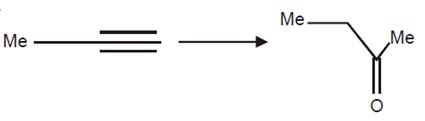The correct statement is
Cyclohexadiene and cyclohexene cannot be isolated with ease during controlled hydrogenation of benzene.
One mole each of benzene and hydrogen when reacted gives 1/3 mole of cyclohexane and 2/3 mole unreacted hydrogen.
Hydrogenation of benzene to cyclohexane is an endothermic process.
It is easier to hydrogenate benzene when compared to cyclohexene.