Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is
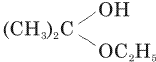
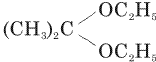
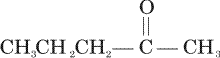
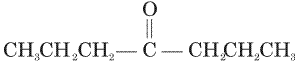

Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is


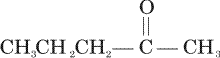
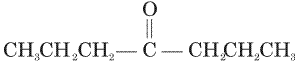
|
Reduction of aldehydes and ketones into hydrocarbons using zinc amalgam and conc. HCl is called |
Answer |
|
$P \underset{2.H_3O^+}{\xrightarrow{\;\;1.CH_3MgBr\;\;}} R \underset{\triangle}{\xrightarrow{\;\;1.dil.NaOH\;\;}}\text{4–methylpent–3–en–2–one}$ |
Answer |
|
Benzaldehyde and acetone can be best distinguished using |
Answer |
|
Which of the following statements is not correct? |
Answer |
|
In which of the following reactions new carbon–carbon bond is not formed |
Answer |
|
Clemmensen reduction of a ketone is carried out in the presence of which of the following? |
Answer |
|
The compound that yields only ketonic compound/s on ozonolysis is |
Answer |
|
The compound obtained when acetaldehyde reacts with dilute aqueous sodium hydroxide exhibits |
Answer |
|
Which of the following has most acidic hydrogen? |
Answer |
|
Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is |
Answer |