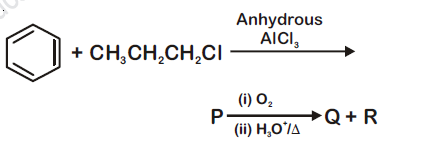The correct order of N-compounds in its decreasing order of oxidation states is
$HNO_3, NH_4CL, NO, N_2$
$HNO_3, NO, NH_4CL, N_2$
$HNO_3, NO, N_2, NH_4CL$
$NH_4CL, N_2, NO, HNO_3$
Explanation:

Additional Questions
|
Which one of the following elements is unable to form $MF_6^{3-}$ ion? |
Answer |
|
Considering Ellingham diagram, which of the following metals can be used to reduce alumina? |
Answer |
|
The correct order of atomic radii in group 13 elements is |
Answer |
|
Which of the following statements is not true for halogens? |
Answer |
|
In the structure of $ClF_3$, the number of lone pair of electrons on central atom ‘Cl’ is |
Answer |
|
Identify the major products P, Q and R in the following sequence of reactions: |
Answer |
|
Which of the following compounds can form a zwitterion? |
Answer |
|
Regarding cross-linked or network polymers, which of the following statements is incorrect? |
Answer |
|
Nitration of aniline in strong acidic medium also gives m-nitroaniline because |
Answer |
|
The difference between amylose and amylopectin is |
Answer |